 Giant covalent structures are insoluble in both polar and non-polar solvents as the covalent bonds in the lattice are too strong to be broken.ĭiamond has a tetrahedral structure held together by strong covalent bonds throughout the structure which makes diamond hard. Giant covalent structures are non conductors of electricity as there are no free charged particles (except in graphite). This is because high temperatures are needed to break the strong covalent bonds within the lattice. This is where atoms are held together by strong covalent bonds in a lattice. Simple molecular structures are soluble in polar solvents as van der Waals' forces are able to form between the polar solvent and the molecular structure. Simple covalent structures are non conductors of electricity because there are no charged particles free to move and conduct. This is because the intermolecular bonds are weak van der Waals' forces so only a small amount of energy is needed to break the bonds. 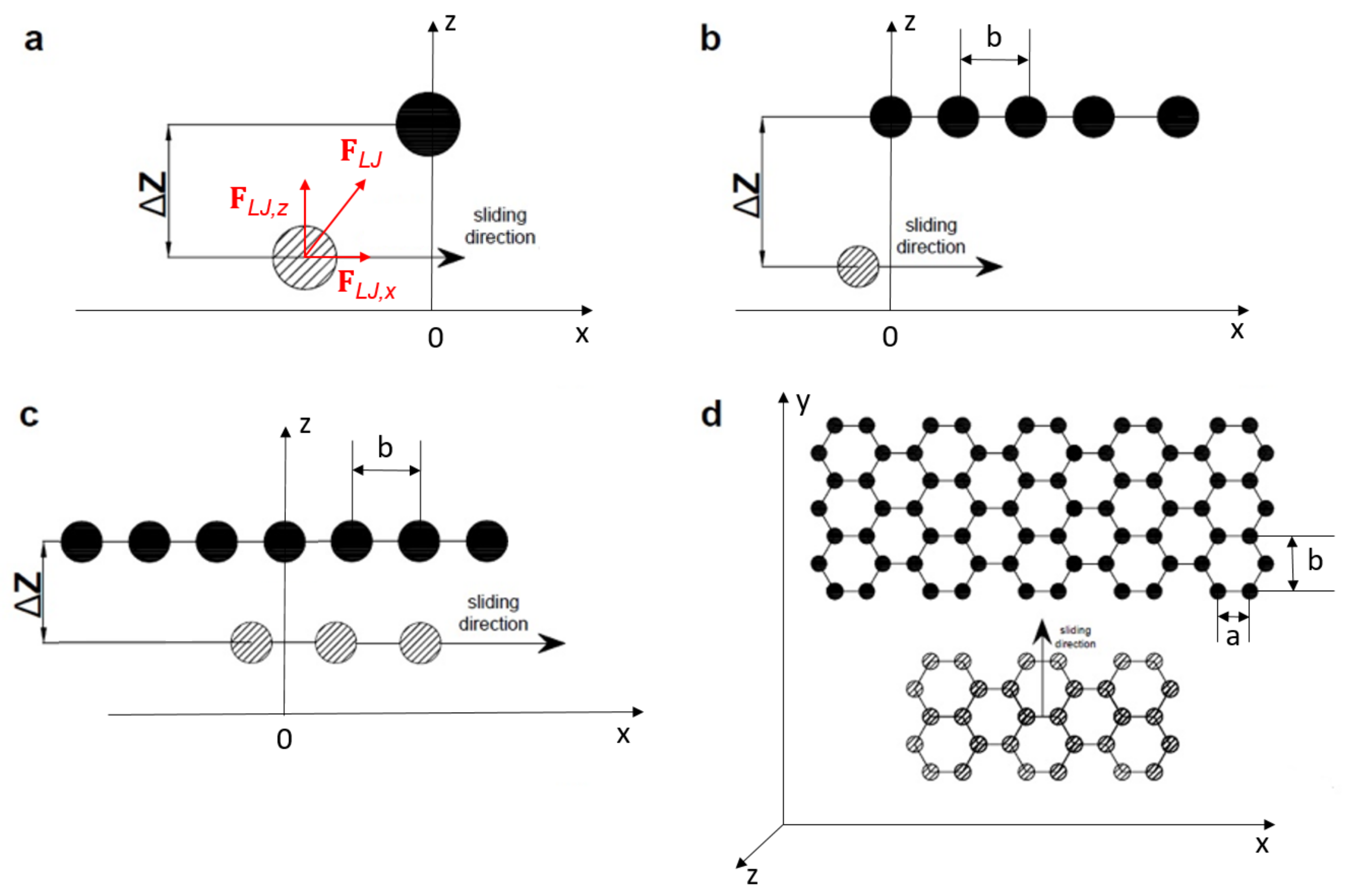
The properties of simple molecular lattices are shown below: Molecules are held together through weak van der Waals' forces. 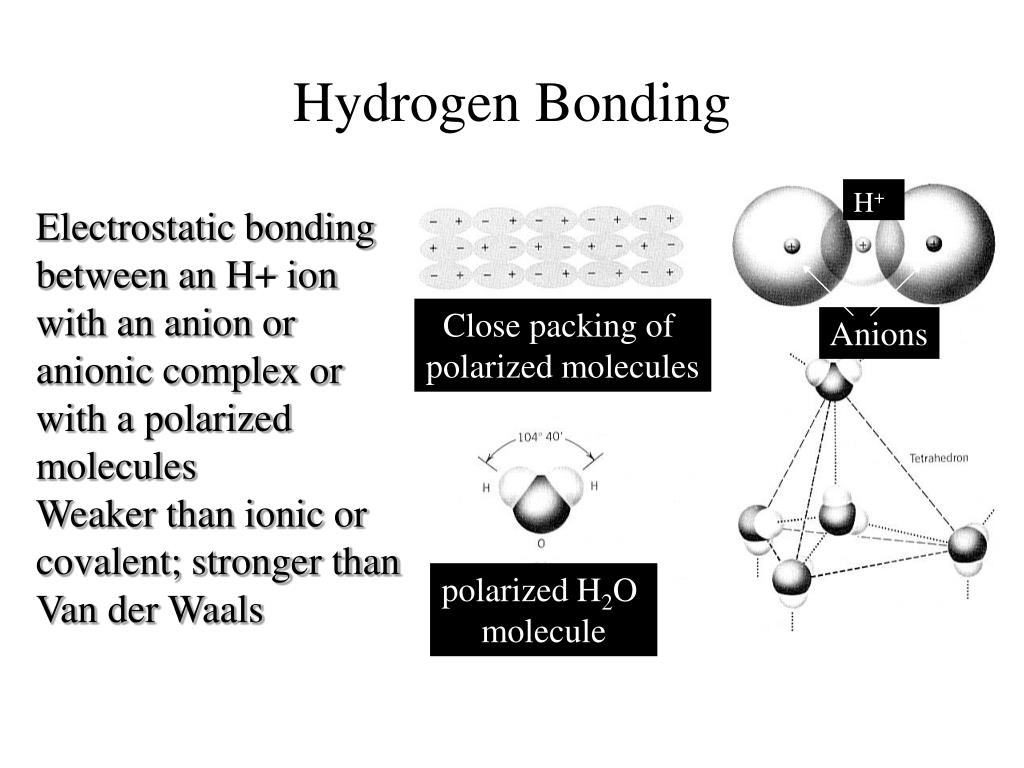
A covalent bond has a shared pair of electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
